A quasi-randomised pilot study on the efficacy and perceived usefulness of adding chemosignals to mindfulness practice for women with social anxiety
This study was carried out within the EU Horizon2020 Project POTION (‘Promoting Social Interaction through Emotional Body Odours’) and conducted at the University of Padua, Italy.
Ethical and safety considerations
The study was conducted in accordance with the declaration of Helsinki, and was approved by the local Ethics Committee, University of Padua (protocol no. 3113). Informed consent was obtained from all participants taking part in the study. A compensation of €13 was provided to participants for their involvement. All study assessments were conducted by trained research staff, and in the case of participants reporting significant mental health symptoms, contact details to relevant help services were provided.
Design
The study utilized a between-subjects design, where participants with SAD underwent two 24-minute mindfulness meditation sessions while allocated to one of four odour conditions: joy BO, fear BO, neutral BO, or clean air. A single-blind procedure was employed, where study participants were blinded to the treatment allocation.
Power-calculation and sample size
An a priori power analysis was conducted based on effect size extrapolated from the previous exploratory study conducted within the POTION project24. The power calculation was performed using G*Power software (version 3.1.9.7) with the function ANOVA, repeated measurements, between factors. The parameters entered were α = 0.05, 1 – β = 0.95, and effect size of between subjects’ analysis of the first trial day of the above-mentioned study (Cohen’s f = 0.3745), for a study design of 4 (odour condition; clean air, neutral BO, fear BO, joy BO) × 2 (time; pre, post). Based on these parameters, a total sample size of 96 was needed.
Body odour collection and Preparation
Sweat samples were collected from a different group of participants, so-called BO donors, at the Instituto Superior de Psicologia Aplicada (ISPA, Lisbon, Portugal). The protocol for sweat collection was approved by the Ethics Committee of ISPA in September 2018. The sweat collection and stimulus composition procedures were similar to those used in our earlier study24, even though additional samples from donors having undergone non-emotional (neutral) emotion inductions were included. Sweat pads used for the current study were based on samples from 26 donors (14 males, mean age = 21.3, SD = 2.5 and 12 females, mean age = 21.6, SD = 3.6). A within -subjects design was used where each BO donor attended the lab at three separate occasions. These sessions were spaced out between days, with the median interval between sessions being 7 days (mean = 8.9), with a minimum of 4 days and a maximum of 27 days between collections. During each session a single emotional state (fear, joy or neutral) was induced, with the order of emotional conditions randomized across sessions. To induce relevant emotional states, donors watched 25-minute video clips that were fear inducing, joy inducing or were non-emotional nature documentaries, whilst sweat was collected via pads placed in the armpits. To ensure successful emotion induction, participants’ emotional states were assessed before and after (for results see supplementary material Table S1). As described above, whilst we decided to use the term “joy BO” instead of “happiness BO”, the change in terminology did not reflect a change in stimulus material, but rather a conceptual re-evaluation of appropriate terminology to describe the condition. Thus the joyful BO stimulus used for the current study was identical to the happiness BO stimulus described in our previous study24.To avoid sweat contamination, participants were asked to undergo a strict behavioural and dietary regimen two days before the body odour collection7. After the emotional induction, pads were removed, frozen at − 80 °C, shipped to the University of Padua (Italy) in dry ice and subsequently stored at − 80 °C. To circumvent potential sex-related, or other influences of individual donors on odour perception43, a super-donor design was used during preparation of the samples for the present study. Each super donor sample included body odour from four different individuals (two males and two females randomly selected, with one male and one female contributing odour samples from the left armpit, and the other male and female from the right armpit), with only one-eighth of collected pad per individual used in the final super donor pool. Similar procedures for collection, storage and preparation have been used and validated in previous studies40,44.
Recruitment and enrolment criteria
Recruitment to the study commenced on the 17/10/2022 and remained open until the recruitment target was set, with the final participant completing the follow-up assessment on the 20/05/2023. Researchers at the University of Padua disseminated the study information via flyers, and individuals expressing an interest were invited to undergo an initial online screening process on the Qualtrics platform. The following inclusion criteria were assessed: (1) female sex, (2) aged 18–35, (3) non-smoker, (4) scoring ≥ 30 on the Liebowitz Social Anxiety Scale in its self-report formulation (LSAS-SR45. The decision to include only women was taken to avoid sex-related confounds regarding olfactory abilities, where women generally outperform men46,47. Participants were ineligible if they: (1) had chronic rhinitis or any other condition potentially impacting odour perception; (2) were pregnant or breastfeeding; (3) reported a diagnosis of mental disorders (including substance abuse disorders) other than social anxiety disorder; (4) reported having severe somatic or neurological conditions; (5) reported current use of psychotropic medications (including antidepressants, antipsychotics, anxiolytics, and mood stabilizers); (6) were presently undergoing psychological therapy; (7) had severe psychotic symptoms (i.e. hallucinations and/or delusions); (8) reported presence of suicidal thoughts or; (9) were unable to provide informed consent.
Participants meeting initial screening criteria were extended invitations for a clinical interview. Within this interview, the presence of social anxiety symptoms was validated using Module F of the Structured Clinical Interview for DSM-5 (SCID-5-CV48, . Furthermore, anosmia or hyposmia were tested using the Sniffin’ Sticks test49 to ensure intact olfactory abilities (for a description see Supplementary Materials). The final study sample included 98 women (fear BO n = 24, joy BO n = 25, neutral BO n = 25, clean air n = 24).
Quasi-randomisation procedure
Allocation of the odour conditions was conducted by research investigators at the site where the study was undertaken (University of Padua), and was done using Excel, creating a list of randomised odour conditions to be used for the study. Allocation to treatment groups occurred in a quasi-randomised manner, as allocation to the odour conditions depended on the day of participation, where those participating on the same day were allocated to the same odour condition (with a maximum of two participants being tested in one day). This procedure was chosen for the pragmatic reason that we aimed to enable optimal use of the sweat pads, given these needed to be discarded after one day of use.
Study procedure and outcome measures
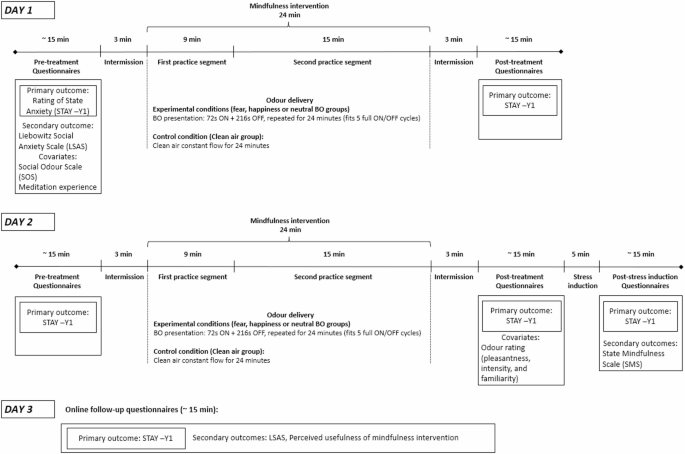
Following informed consent and screening, participants were assigned to one of the four odour conditions in which they remained throughout all experimental sessions. The intervention was performed over two consecutive days, with the experimental procedure, including data collection and mindfulness practice, lasting approximately 60 min per day. One hour prior to each session, participants were instructed to abstain from consuming food and beverages, excluding water. Figure 1 outlines the study procedure and questionnaires collected at each time-point.
Study intervention and stress-induction
The intervention took place at the University of Padua. The mindfulness training was done individually and performed using two mindfulness practices presented through recorded audio tracks available from a smartphone app called “Con tatto” (developer LifeSTech research team). The practices focussed on conscious breathing, meditation and relaxation exercises. The first practice segment (“The breath that frees”) was 9 min long whereas the second practice (“The thin breath”) was 15 min long. Throughout the exercises participants were asked to be present in the moment, and to observe their bodily sensations and their breathing. All participants listened to the guided mindfulness practice via noise cancelling headphones. The total length of the practice was 24 min. Throughout the mindfulness practice, BOs or clean air were delivered with a custom-built50 continuous airflow, computer-controlled olfactometer, directly to both nostrils with a nasal cannula during the intervention. Airflow was kept constant between 50 and 70 ml/min, BOs were delivered in 72 s-long pulses separated by a 216 s clean air. As indicated in Fig. 1, following the mindfulness session on day 2, participants underwent a stress-induction where they were given an abstract, and told they needed to give a presentation about the abstract to a small group of people, which they were given 3 min to prepare for. State anxiety levels were assessed before and after the stress-induction. At the end of the experimental session on day 2, participants were told that the presentation had been cancelled, and they did not need to present.
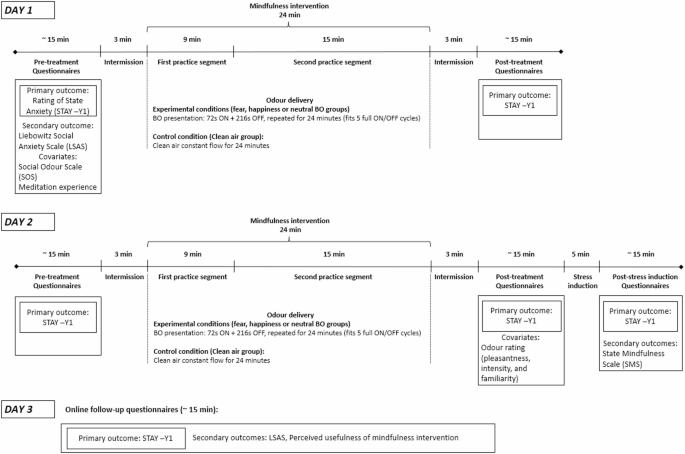
Schematic outline of the study procedure.
Primary outcome
State-Trait Anxiety Inventory (STAI)51: A 40-item self-report scale that assesses two forms of anxiety, state anxiety (STAI-Y1) and trait anxiety (STAI-Y2), 20 items each, on a four-point Likert scale (ranging from 1 (“not at all” in the State subscale STAI-Y1 and “almost never” in the Trait subscale STAI-Y2) to 4 (“very much” in the State subscale STAI-Y1 and “almost always” in the Trait subscale STAI-Y2), where higher scores indicate elevated levels of anxiety. For the current study, the 20-item STAY-Y1 scale was used, as we were interested in ratings of state anxiety levels. Scores range from 20 to 80, cutoffs are typically 20–39 for no or low anxiety, 40–59 for moderate anxiety and 60–80 for high anxiety51.
Secondary outcomes
Perceived usefulness: This was rated with one item asking participants how useful they thought the mindfulness sessions were and rated from 0 (“not useful at all”) to 10 (“very useful”).
State Mindfulness Scale (SMS)52: A 21-item self-report questionnaire assessing two state mindfulness aspects: one measuring state mindfulness of bodily sensations and the other reflecting state mindfulness of the mind, using a 5-point Likert scale from 1 (not at all) to 5 (very well), where a high score reflects a high state of mindfulness.
The Liebowitz Social Anxiety Scale (LSAS-SR)45: A 24-item questionnaire where fear and avoidance in a range of social and performance situations are assessed. It is rated on a 4-point scale, where a higher score reflects higher anxiety.
Covariates
Social Odor Scale (SOS)53: A 12-item self-report questionnaire designed to measure body odour awareness, including individual body odour, familiar and unfamiliar body odours, romantic partner body odour, and body odour of strangers. Items are rated on a 5-point scale, where a high score indicates higher body odour awareness.
Odour ratings: Subjects were asked to rate the intensity, pleasantness, and familiarity of the odour presented during the mindfulness intervention, each on a 10-point scale, where a higher score indicated higher odour intensity, pleasantness, and familiarity. Similar rating procedures have been used in previous olfaction studies (e.g54. To avoid drawing participants attention towards specific aspects of the olfactory content of the experiment as it was ongoing, questions regarding odour ratings were given after the experimental sessions were completed on day 2.
Prior meditation experience: To control for prior meditation experience the question “Do you practice, or have you ever practiced mindfulness meditation?” was asked to everyone. If yes, there was a follow-up question “how often do you practice?” with possible answers “not currently”, “practice less than one time a week”, “less than three times a week”, “practice three times a week or more”, “practice every day”.
Trial registration
With recruitment commencing on the 17/10/2022, the trial was retrospectively registered on date: 16/11/2022 (ISRCTN Registry No. 98675422). The registration also included investigating physiological responses to BOs as secondary outcomes, results which will be reported in a separate publication. Due to unforeseen recruitment challenges, mainly brought on by the COVID-19 pandemic, screening criteria was made less stringent after study commencement, to increase the potential recruitment pool. This change was implemented on 19/12/2022 and entailed alterations to the LSAS screening threshold from ≥ 50 to ≥ 30. Whilst a score of 50 may have lessened the risk of floor effects, this reduction was deemed necessary to ensure timely recruitment. Importantly, the presence of SAD was still validated using Module F of the Structured Clinical Interview for DSM-5 (SCID-5-CV48. Similar LSAS thresholds have been set in other studies alleviating symptoms of social anxiety (e.g55).
Statistical analysis
Data were analysed using R statistical software (version 4.1). Differences in demographic data, baseline characteristics and odour ratings were explored by means of one-way ANOVA or Kruskal-Wallis test when normality was violated. For descriptive purposes, the magnitude of change (mean difference and Cohen’s d) of pre- to post-treatment changes within groups, as well as between-groups comparisons, were assessed. These analyses were conducted separately for Day 1 and Day 2, as well as for the change from pre-treatment on Day 1 to follow-up on Day 3. Effect sizes were interpreted using conventional thresholds: d < 0.2 as negligible, 0.2 ≤ d < 0.5 as small, 0.5 ≤ d < 0.8 as medium, and d ≥ 0.8 as large56. Pairwise t-tests with Holm correction for multiple comparisons were employed to evaluate these changes. In addition, whilst the sample met diagnostic thresholds for social anxiety, the majority of participants were in the low to moderate range of state anxiety (STAI-Y1) at baseline. Given this baseline profile, we chose not to rely on severity categories for interpreting treatment effects. Instead, Jacobson & Truax57 method was used to assess whether STAI-Y1-score changes reached clinically meaningful levels.
Test of primary hypothesis
To examine the primary hypothesis, changes in state anxiety (STAI-Y1) scores were calculated through linear mixed modelling, where a 4 (Odour condition) × 2 (Day) × 2 (Time) model was tested. Odour condition (clean air vs. fear BO vs. joy BO vs. neutral BO) served as the between-subject factor, while time (pre-treatment vs. post-treatment) and day (day 1 and 2) were within-subject factors. Statistical models were applied through the lme4 and lmerTest R packages. To control for variations in pre-treatment state anxiety scores, all models were fitted using random intercepts.
Secondary outcomes
Secondary outcomes (change following stress-induction, change at follow-up, perceived usefulness and state mindfulness scores) were explored by means of one-way ANOVA or Kruskal-Wallis test when normality was violated. Changes in STAI-Y1 scores after the stress induction and changes from pre-treatment on day 1 to the final follow-up assessment point on day 3 were examined through linear mixed modelling.
Covariates
Analyses were conducted with and without covariates. Because there were fewer data points for the SOS scale due to this scale being introduced at a later stage during the trial (data collected from 70 participants; clean air n = 16, fear n = 14, joy n = 17 neutral n = 23), three separate models were tested; one without the planned covariates, that included the full sample (Model 1), one where odour ratings and prior meditation experience were added as covariates (Model 2) and one where odour ratings and prior meditation experience and the SOS were included as covariates (Model 3). The outputs of all models are presented in the supplementary materials, and unless the addition of covariates substantially influenced the main results, the output of the analyses without the covariates are reported in the main text, as these analyses comprised the full sample.
link







